Camptothecin
Camptothecin has a planar pentacyclic ring structure, that includes a pyrrolo[3,4-β]-quinoline moiety (rings A, B and C), conjugated pyridone moiety (ring D) and one chiral center at position 20 within the alpha-hydroxy lactone ring with (S) configuration (the E-ring). Its planar structure is thought to be one of the most important factors in topoisomerase inhibition.
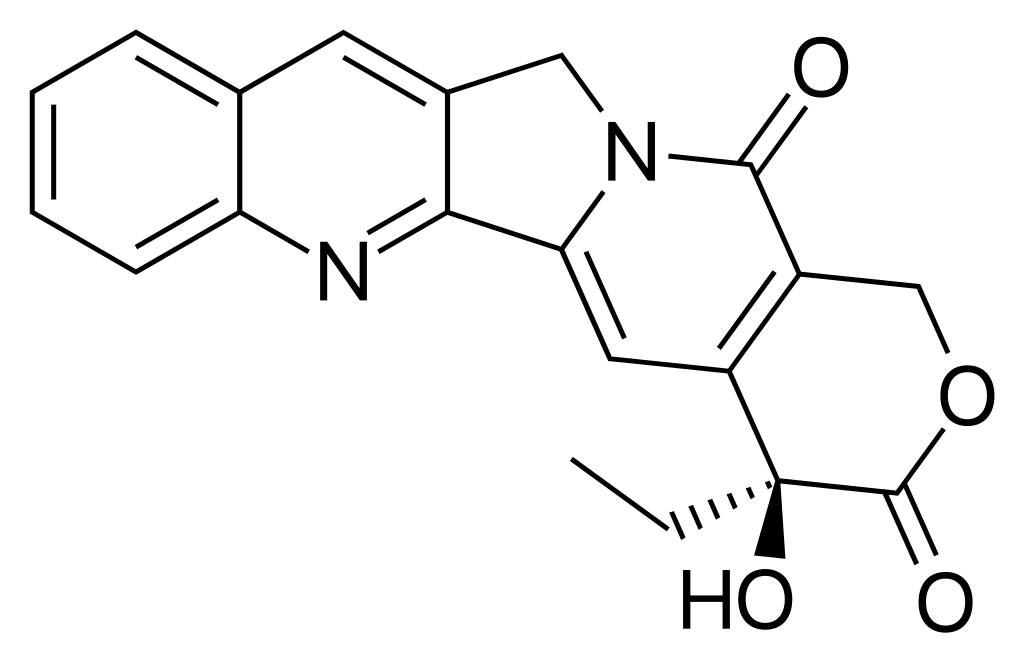
CPT makes a ternary complex between DNA and topoisomerase II.

Interactions with the target
PDB 1T8I
Questions
- Provide additional examples of a small molecule acting as a “glue” bringing together two macromolecules such as two proteins or a protein and a nucleic acid.